Novo Nordisk stumbles again: CagriSema trails Lilly’s Zepbound in key trial
-
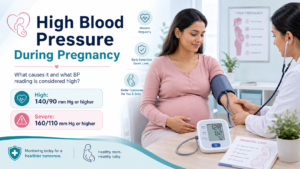
Novo said its next-gen obesity drug CagriSema hit 23% weight loss at 84 weeks, but it failed to prove non-inferiority versus Lilly’s tirzepatide (Zepbound) at 25.5%.
-
Novo stock dropped about 15–16% on the day across reports; several outlets also cited a steep 12-month decline.
-
Lilly raised the convenience bar by rolling out a single Zepbound pen with four doses (one month).
-
Novo defended the result as meaningful; the company signaled more trials and awaits an FDA decision expected in late 2026.
-
Investors and analysts talked about strategy shifts, including diversification and M&A themes, after the miss.
What is the main takeaway from the CagriSema news?
CagriSema lost the headline comparison on weight loss versus Zepbound. Novo reported 23% vs 25.5% at 84 weeks, and that gap mattered because the study’s primary goal was a non-inferiority claim. Next, the numbers need clear context.
What did Novo actually report in the late-stage trial?
Novo reported 23% average weight loss at 84 weeks for CagriSema and said it did not meet the primary endpoint versus Lilly’s tirzepatide result of 25.5% at the same time point (as described in the coverage you shared). Next, it helps to see the comparison in one view.
CagriSema vs Zepbound (as reported)
| Item | Novo Nordisk | Eli Lilly |
|---|---|---|
| Drug in the headline comparison | CagriSema | Tirzepatide (Zepbound) |
| Reported weight loss | 23% | 25.5% |
| Reported duration | 84 weeks | 84 weeks |
| Primary trial goal mentioned | Non-inferiority vs comparator | Reference comparator result |
Next up: what “non-inferiority” means in plain English.
What does “non-inferiority” mean here?
Non-inferiority means Novo tried to show CagriSema is not meaningfully worse than Zepbound on weight loss. Novo said it missed that bar. That single label can change how payers, doctors, and investors rank a drug. Next: why the market reacted so hard.
Why did Novo’s shares drop after the announcement?
The market treated the miss as a competitive signal. When a “next-generation” candidate fails to match the leader, investors often cut expectations for future share, pricing power, and growth. Reports you shared cited a ~15–16% one-day drop. Next: Lilly’s move on convenience.
What changed with Lilly’s new Zepbound pen?
Lilly said Zepbound is now available as one pen containing four doses. Coverage framed it as a simpler monthly routine because patients use fewer devices. Convenience can influence adherence and preference, even when efficacy headlines dominate. Next: Novo’s response.
How did Novo frame a 23% weight-loss result?
Novo called 23% “significant” and said it was pleased with the outcome. In the coverage, Novo’s chief scientific officer highlighted the clinical meaning of the loss even without winning the direct comparison. Next: a key design detail raised in the reporting.
Did the trial design matter in how people read the results?
Yes. The coverage described the study as open-label. An open-label design means participants know which treatment they receive, which can introduce bias risks in comparisons. That nuance can affect how confidently people interpret a small gap. Next: what this means for the broader GLP-1 race.
Why does this matter in the Novo vs Lilly GLP-1 rivalry?
GLP-1 obesity drugs are one of pharma’s most valuable battlegrounds. Novo built early dominance with semaglutide brands like Wegovy and Ozempic. The reporting said Lilly has pulled ahead in prescriptions and market share in the U.S. Next: the extra headwinds Novo is managing.
What other pressures are hitting Novo, according to the reports?
The coverage linked the setback to broader strain: competition intensifies, U.S. pricing faces pressure, and some markets approach exclusivity changes for legacy brands. The reporting also discussed copycat compounding and regulatory scrutiny around replicas. Next: what investors are pushing Novo to do.
What are investors and analysts asking Novo to do now?
Some investors want a pivot and more diversification. The Bloomberg excerpt you shared described calls for Novo’s CEO to broaden beyond diabetes and obesity dependence. The CNBC excerpt cited an analyst talk about M&A needs and large potential spend figures. Next: what Novo can do with CagriSema from here.
What can Novo do next with CagriSema?
Novo can still compete by expanding evidence and sharpening positioning.
-
Run additional trials (including higher-dose combinations mentioned in the coverage).
-
Complete FDA review (an FDA decision was described as expected late 2026).
-
Differentiate on profile if later data show advantages beyond average weight loss (example: usability, tolerability, specific patient segments).
References:
https://www.cnbc.com/2026/02/23/novo-nordisk-stock-cagrisema-trial-fails-weight-loss.html
https://www.axios.com/2026/02/23/ozempic-novo-nordisk-eli-lilly-zepbound